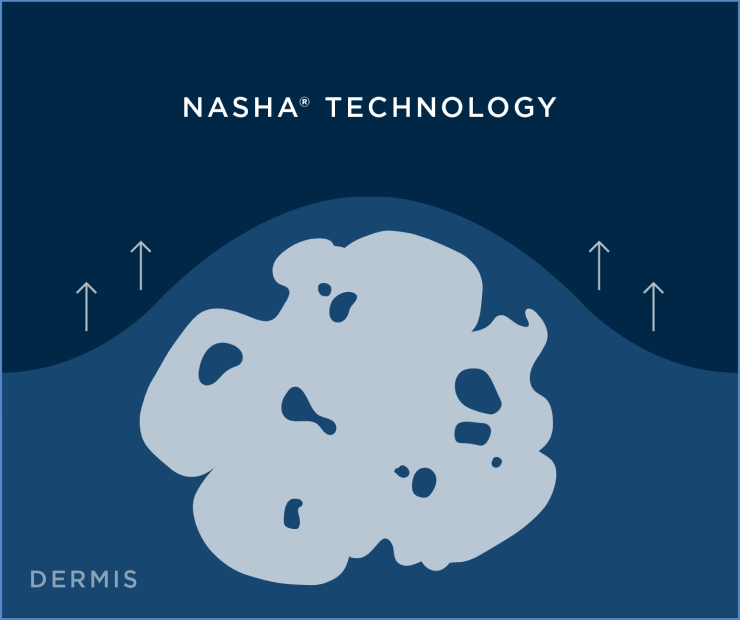
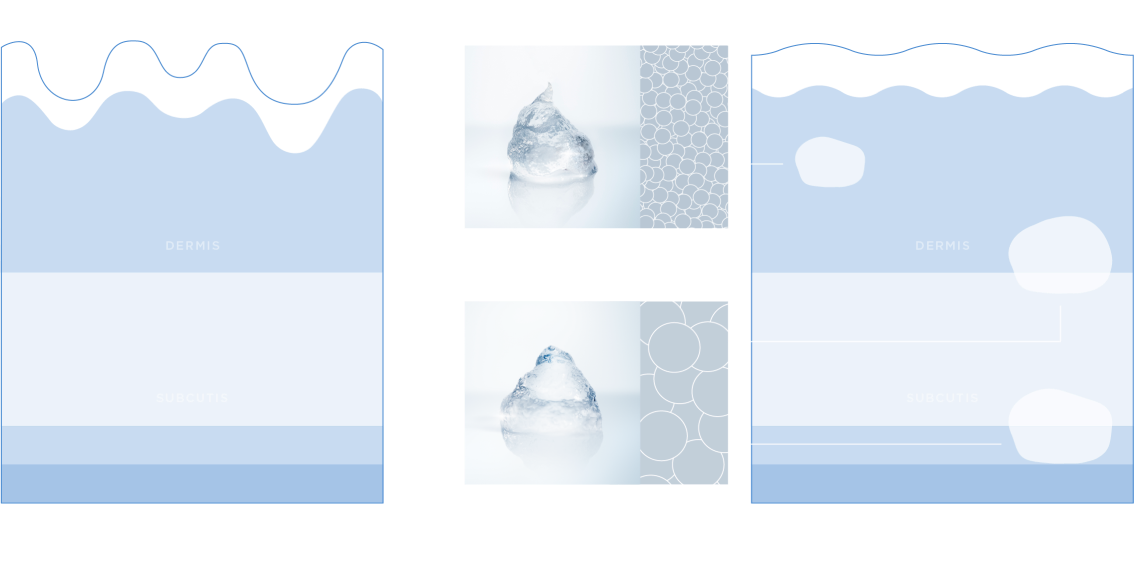
Restylane and Restylane-L are for mid-to-deep injection into the facial tissue for the correction of moderate-to-
severe facial wrinkles and folds, such as nasolabial folds, and for lip enhancement.
Restylane Lyft with Lidocaine is for deep implantation into the facial tissue for the correction of moderate-to-severe facial wrinkles and folds, such as nasolabial folds, and for cheek augmentation and for the correction of age-related midface contour deficiencies. Restylane Lyft with Lidocaine is also indicated for injection into the dorsal hand to correct volume loss. Restylane Lyft with Lidocaine is also indicated for injection into the mid-to-deep dermis (subcutaneous and/or supraperiosteal) for augmentation of the chin region to improve the chin profile in patients with mild-to-moderate chin retrusion.
Restylane Silk is for lip augmentation and for correction of perioral wrinkles.
Restylane Kysse is for lip augmentation and for correction of upper perioral wrinkles.
Restylane Refyne is for mid-to-deep injection into the facial tissue for the correction of moderate-to-severe facial wrinkles and folds, such as nasolabial folds.
Restylane Defyne is for mid-to-deep injection into the facial tissue for the correction of moderate-to-severe, deep facial wrinkles and folds, such as nasolabial folds. Restylane Defyne is also indicated for injection into the mid-to-deep dermis (subcutaneous and/or supraperiosteal) for augmentation of the chin region to improve the chin profile in patients with mild-to-moderate chin retrusion.
Restylane Contour is for cheek augmentation and for the correction of midface contour deficiencies, and for correction of temple hollowing.
Restylane Eyelight is for the improvement of infraorbital hollowing.
Do not use if you have severe allergies that have required in-hospital treatment, are allergic to lidocaine or gram-positive bacterial proteins used to make hyaluronic acid, prone to bleeding, or have a bleeding disorder. The safety of use while pregnant or breastfeeding has not been studied. Tell your doctor if you are planning other cosmetic treatments (such as lasers and chemical peels) as there is a possible risk of inflammation at the injection site.
The most common side effects include swelling, redness, pain, bruising, headache, tenderness, lump formation, itching at the injection site, and impaired hand function (Restylane Lyft – hand injection). Tell your doctor if you’re taking medications that lower your body’s immune response or affect bleeding, such as aspirin or warfarin; these medications may increase the risk of bruising or bleeding at the gel injection site. Use at the site of skin sores, pimples, rashes, hives, cysts, or infection should be postponed until healing is complete.
Delayed-onset inflammation near the site of dermal filler injections is one of the known adverse events associated with dermal fillers, and cases have been reported to occur at the dermal filler treatment site following viral or bacterial illnesses or infections, vaccinations, or dental procedures. Typically, the reported inflammation was responsive to treatment or resolved on its own. Serious but rare side effects include delayed-onset infections, recurrence of herpetic eruptions, superficial necrosis, and scarring at the injection site. The risk of unintentional injection into a blood vessel is small but can occur and could result in serious complications, which may be permanent including, vision abnormalities, blindness, stroke, temporary scabs, or permanent scarring of the skin. As with all skin injection procedures, there is a risk of infection.
To learn more about serious but rare side effects and full Important Safety Information, visit
www.RestylaneUSA.com.